Refining the route to solubility: Publication showcases a new FEP+ method to predict solubility prospectively in live drug discovery projects
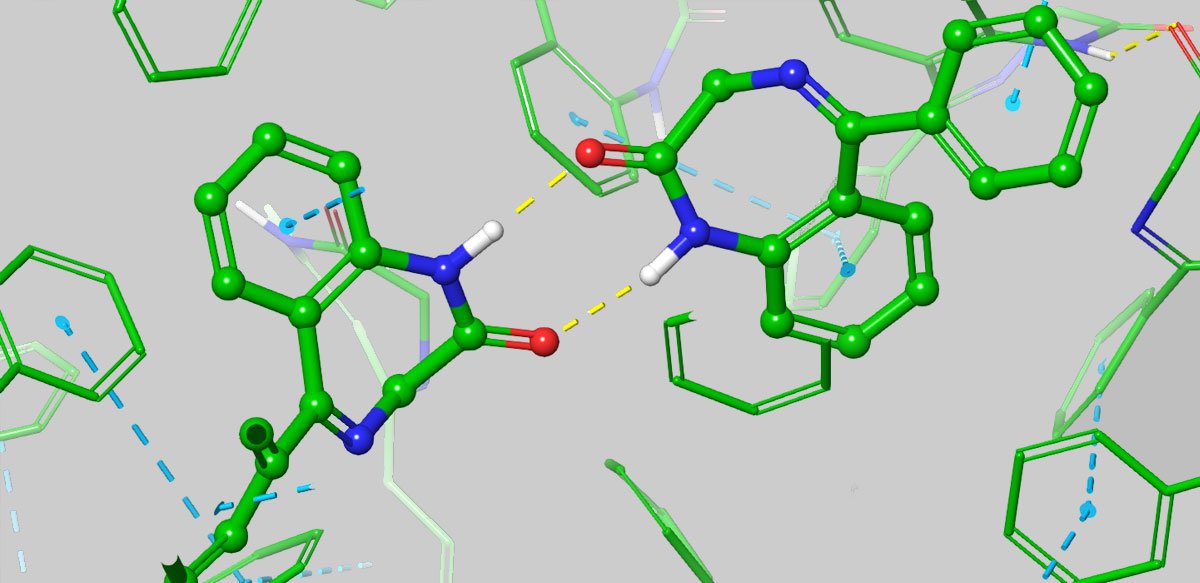
In collaboration with Janssen and Nimbus, Schrödinger published a preprint in ChemRxiv, A Free Energy Perturbation Approach to Estimate the Intrinsic Solubilities of Drug-like Small Molecules. The paper presents a novel physics-based approach to predicting the aqueous solubility of small molecules that takes into account three-dimensional solid-state characteristics in addition to polarity. The method performed well not only on public datasets, but in testing against the measured solubilities from a large number of advanced programs in-house at Janssen. Furthermore, the paper reports on the first prospective application of the method in a live drug discovery project in collaboration with Nimbus Therapeutics.
Bringing together FEP+ Solubility, FEP+ potency, RRCK passive permeability modeling, and physico-chemical parameters like PSA in a modeling screening funnel can substantially improve the probability of success and accelerate the project timeline.
Dr. Mondal explains the paper’s key findings:
Tell us what’s the key advance in this paper, and why is it important?
In this paper, we introduce a brand new technology, FEP+ Solubility—a physics-based method that provides a detailed examination of the 3D solid-state packing structure of a molecule and accurately predicts solubility without a training set. FEP+ Solubility is built on our free energy perturbation method, FEP+, and is both accurate and has a wide domain of applicability, making it particularly suitable for drug discovery research, where exploring novel chemical space could be crucial to success.
Aqueous solubility is a focal point during lead optimization in drug discovery because it affects bioavailability, a molecule’s ability to enter the bloodstream and circulate throughout the body. Given that it is estimated that almost 40% of all new chemical entities (NCE) are determined to be insoluble, or nearly so, and that molecules with low solubility often experience issues during clinical and pre-clinical trials, leading to failure, it becomes clear why improving solubility is often a focal point during lead optimization. Moreover, straightforward attempts to increase solubility during lead optimization can degrade membrane permeability and/or potency.
How did researchers traditionally try to improve solubility? And how does FEP+ Solubility compare with previous approaches?
The most common approach to improving solubility was empirical—guided usually by logP or sometimes by machine learning models fitting existing data of experimentally measured solubility to various molecular descriptors, such as logP and polar surface area. However, such empirical approach can actually lead one astray, for example, simply adding polar groups can preferentially stabilize the solid state, and lead to the sometimes counterintuitive result of reducing solubility. FEP+ Solubility, on the other hand, allows for a detailed computation of the 3D solid-state packing energetics, providing insightful predictions about interactions that can improve or degrade solubility. A great example of this is Benzodiazepine, where it has been shown that substitutions that disturb favorable solid-state interactions can lead to improved solubility.
Another key difference between empirical methods and FEP+ Solubility is that the former requires a training set, and thus is inherently limited in its applicability to the chemical space of the training set, and often struggles to extrapolate to novel chemical entities. Unfortunately, it’s often necessary to expand beyond this limited scope of chemical space in order to achieve the desired solubility in balance with other parameters or IP, and this is where FEP+ Solubility really shines with its large domain of applicability. Furthermore, the understanding gained from FEP+ Solubility studies such as elucidating an in silico solubility SAR, can help the project team ideate novel chemical structures to improve solubility.
Can you speak to the performance of this new method, FEP+ Solubility, and how you are using the technology?
Sure, we did extensive retrospective studies to test our model against available published solubility data for compounds of pharmaceutical interest, and saw great agreement. But more importantly, we began to use the model in prospective studies, and again we saw excellent performance by FEP+ Solubility in classifying compounds based on their solubility profile, making it a quotidian tool in our arsenal.
As part of Schrödinger’s Drug Discovery Group, we use the Schrödinger platform in all of our internal and collaborative drug discovery projects. The platform consists of three things: advanced physics-based methods, machine learning, and enterprise informatics.
The predictions from various physics-based methods including FEP+ Solubility and machine learning methods, where applicable, are captured within LiveDesign, our web-based enterprise informatics program, facilitating synthesis decisions based on a holistic comparison of a large number of ideated molecules. The most promising compounds emerging from each round are then further optimized through additional cycles of computational analysis enabling us to determine the best candidates for synthesis.
FEP+ Solubility sounds like a significant advancement, how do you see it impact drug discovery projects?
Drug discovery teams often find themselves trapped in lengthy and uncertain cycles of optimization where they change R-groups to improve solubility, only to lose ground against potency, or an ADME property like permeability; or vice versa. These parameters need to be satisfied in the same molecule but are generally anti-correlated, meaning that if we are limited to designing along the dimension of polarity / hydrophobicity alone, often it would be to the detriment of other important properties.
By going beyond just polarity, it becomes easier to overcome the potency / selectivity / permeability / solubility tradeoffs to obtain higher quality chemical matter faster. With FEP+ Solubility used in conjunction with FEP+ analyses of potency and selectivity, and RRCK passive permeability modeling at scale, it becomes possible to break out of insoluble regimes and achieve balanced profiles within 1-2 cycles of synthesis.
This is especially true for projects where most synthesized compounds are found to be insoluble, the probability of success for meeting the Development Candidate guidelines can be dramatically improved by focusing synthesis on high quality chemical matter predicted to be soluble, potent, selective, and permeable.
The value of using FEP+ for solubility is particularly high when using it together with FEP+ for potency and selectivity, and RRCK permeability modeling, because the net outcome is more than the sum of the individual analysis. For example, one may decide to remove an aromatic ring based on FEP solubility results; however, if the aromatic ring is productive in the binding site, the probability of success is much higher if FEP+ potency modeling is used to maintain the potency while replacing the aromatic ring.
Our modeling platform allows researchers to map out the SAR for potency, selectivity, permeability, and solubility side-by-side for different series and sub-series. This information can help teams strategize which core to prioritize for further optimization, how to functionalize a given core, and to stimulate the iterative design of new ideas.
Any final thoughts?
To summarize, our approach through the FEP+ framework provides an opportunity to systematically predict the solubility of novel molecular entities. Beyond early stage drug discovery, this work lays down the foundation for solving key problems in formulation and materials science contexts with computational modeling. That is a world of impactful advances waiting to be made!
